Metallic zirconium is a high-performance, silvery-grey transition metal renowned for its exceptional corrosion resistance, high ductility, and specialized role in nuclear engineering. Many industrial procurement managers struggle to balance material durability with cost-efficiency, often facing premature equipment failure in aggressive chemical environments. You can solve these engineering bottlenecks by understanding the nuanced performance of zirconium vs titanium in high-stress applications.
What defines metallic zirconium and how does zirconium vs titanium vary?

Metallic zirconium is defined by its atomic number 40 and its unique ability to remain stable under extreme thermal and chemical duress. When evaluating zirconium vs titanium , engineers note that while both belong to the same periodic group, zirconium offers superior resistance to specific mineral acids and alkalis. Its visual luster is often described as golden-grey, distinguishing it from the purely silver sheen of other industrial metals.
Historical discovery and isolation
The German chemist Martin Heinrich Klaproth first identified the element in 1789 while analyzing gemstone samples. However, the metal was not isolated in its pure, high-ductility form until the mid-1920s. This delayed adoption was primarily due to the difficulty of removing impurities like nitrogen and oxygen.
Chemical classification and atomic data
Zirconium occupies the fourth group of the fifth period in the periodic table. It possesses high plastic properties, allowing it to be forged or rolled into complex shapes. Think about it. This atomic structure is what grants the metal its remarkable paramagnetism and heat stability.
- Atomic Number: 40
- Appearance: Silvery-grey with a yellowish tinge
- Crystal System: Hexagonal close-packed (at room temperature)
- Hardness: 640-670 MN/m² (Brinell)
Key Takeaway: Understanding the chemical foundations of zirconium allows you to predict its behavior in metallurgical alloying and precision engineering.
| Property | Value | |
|---|---|---|
| Atomic Weight | 91.224 | |
| Density | 6.52 g/cm³ | |
| Electrical Resistivity | 42.1 μΩ·cm | |
| Thermal Conductivity | 22.7 W/(m·K) |
This fundamental data suggests that zirconium is an ideal candidate for applications where thermal expansion must be tightly controlled.
What raw materials are used for zirconium vs titanium production?

Zirconium is primarily extracted from zircon (zirconium silicate) and baddeleyite minerals found in coastal sands and igneous rock formations. The sourcing process for zirconium vs titanium differs significantly, as zirconium is never found in its pure metallic form in nature. Instead, it exists as a trace element within the lithosphere, requiring complex multi-stage purification to reach industrial grades.
Primary zirconium-bearing minerals
Zircon remains the most vital raw material for the industry, often found in granites and syenites. Baddeleyite and eudialyte are also significant sources, though they are less widely distributed. Here is the deal: the purity of these ores determines the final quality of the metallic ingot.
Global distribution of ore deposits
Explored reserves of zirconium minerals are estimated at approximately 45 million tons worldwide. Major deposits are concentrated in Australia, South Africa, and India, which together dominate the global supply chain. You should know that Australia and South Africa alone provide nearly 60% of the world’s zircon concentrate.
- Australia: Leading global producer of high-grade zircon sand.
- South Africa: Significant source of baddeleyite and industrial concentrates.
- USA: Holds reserves of over 7 million tons.
- China: Major processor of raw concentrates into metallic products.
Key Takeaway: A diversified sourcing strategy is essential for you to mitigate supply chain risks associated with geographic concentration in ore mining.
| Major Producer | Est. Reserves (Million Tons) | Share of Global Production (2020) | |
|---|---|---|---|
| Australia | 14 | ~30% | |
| South Africa | 11 | ~25% | |
| India | 27 | ~10% | |
| Others | 8 | ~35% |
The concentration of reserves in a few key nations underscores the importance of establishing long-term partnerships with reliable material suppliers.
Where is the industrial demand for zirconium vs titanium growing?

Industrial demand for zirconium is expanding rapidly across the chemical, aerospace, and energy sectors due to its niche performance advantages. When comparing the growth of zirconium vs titanium, the zirconium vs titanium decision often hinges on specific resistance to hydrochloric or sulfuric acids. While titanium is a versatile workhorse, zirconium is the preferred choice for the most aggressive “mission-critical” environments.
Heavy industry and machine building
Zirconium is utilized as a structural material for manufacturing acid-resistant chemical reactors and high-pressure pumps. Its ability to withstand high temperatures makes it indispensable for components that operate in continuous-duty cycles. But that’s not all. It serves as a vital alloying additive that increases the mechanical strength of other metals.
Precision aerospace components
In jet engines and spacecraft, zirconium alloys provide the heat resistance necessary for supersonic travel. These materials are often alloyed with niobium or tin to enhance creep resistance. What does this mean for you? It means your components can operate longer at higher temperatures without losing structural integrity.
- Jet Engines: Used in turbine blades and heat shields.
- Rocketry: Indispensable for nozzles and combustion chambers.
- Abrasives: Zirconium compounds create high-performance grinding media.
- Pyrotechnics: Used for producing bright, smokeless fireworks.
Key Takeaway: Identifying the specific environmental stressors of your project will help you decide if zirconium’s premium performance justifies the investment.
| Industry Sector | Primary Application | Key Benefit | |
|---|---|---|---|
| Chemical Processing | Acid-resistant reactors | Zero corrosion in HCL | |
| Aerospace | Propulsion components | High-temp stability | |
| Metallurgy | Alloying additive | Enhanced grain structure | |
| Ceramics | Opacifiers | High whiteness and opacity |
This cross-sector utility demonstrates that zirconium is no longer a niche metal but a foundational element of modern industrial infrastructure.
Why is corrosion resistance better for zirconium vs titanium?

Zirconium offers superior corrosion resistance because it forms a dense, self-healing oxide layer that is more stable than that of titanium in many organic and inorganic acids. In the debate of zirconium vs titanium, zirconium stands out for its total immunity to corrosion in most concentrations of hydrochloric acid and alkaline solutions. This makes it the gold standard for equipment that must endure decades of exposure to harsh chemical reagents.
Stability in acids and alkalis
The metal does not react with sulfuric acid or common alkalis even at elevated temperatures. Unlike other metals that might pit or scale, zirconium maintains a smooth surface finish in the presence of chlorides. Believe it or not, it remains unaffected by seawater and chlorinated water, preventing the biofouling common in marine hardware.
Protective oxide film formation
The secret to its longevity lies in the spontaneous formation of zirconium dioxide (ZrO₂) on the surface. This film is incredibly hard and adheres tightly to the substrate metal. Look at this: even if the surface is scratched, the oxide layer reforms instantly in the presence of oxygen or moisture.
- Self-Healing: Instant restoration of the protective barrier.
- Pitting Resistance: High tolerance for chloride ions.
- Thermal Stability: The oxide film remains intact even at several hundred degrees.
Key Takeaway: Choosing zirconium for acid-handling systems can virtually eliminate the downtime and replacement costs associated with chemical corrosion.
| Corrosive Media | Zirconium Performance | Titanium Performance | |
|---|---|---|---|
| Hydrochloric Acid | Excellent (up to 37%) | Limited at high concentrations | |
| Nitric Acid | Excellent | Good | |
| Sodium Hydroxide | Outstanding | Moderate | |
| Seawater | Immune | Excellent |
The technical superiority of zirconium in alkaline and specific acidic environments provides a significant safety margin for chemical plant operations.
How do thermal limits compare for zirconium vs titanium use?
Zirconium possesses a significantly higher melting point than titanium, making it more suitable for extreme high-temperature industrial processes. When evaluating zirconium vs titanium , the melting point of zirconium is approximately 1855°C, while titanium melts at a lower 1668°C. This thermal gap is crucial for vacuum furnace components and structural elements in high-energy physics.
Melting and boiling point analysis
With a boiling point of 4409°C, zirconium is one of the most stable elements under vacuum distillation. It preserves its high-quality mechanical properties at both ends of the temperature spectrum. It gets even better: zirconium dioxide is one of the most refractory substances on Earth, widely used in kiln linings.
Low-fusible dioxide compounds
While the metal is ductile, its oxide form (zirconia) is prized for its thermal shock resistance. This allows for the production of crucibles and foundry molds that can withstand the pouring of molten steel. Want to know more? Zirconium’s low thermal expansion coefficient ensures that parts remain dimensionally stable during rapid heating and cooling cycles.
- Refractory Use: Ideal for glass melting tanks.
- Foundry: Excellent for investment casting molds.
- Grinding: High density (>6.0 g/cm³) ensures efficient milling at high speeds.
Key Takeaway: You can push the boundaries of your thermal processing limits by incorporating zirconium alloys into your high-heat assemblies.
| Metal | Melting Point (°C) | Boiling Point (°C) | |
|---|---|---|---|
| Zirconium | 1855 | 4409 | |
| Titanium | 1668 | 3287 | |
| Zirconium Dioxide | ~2715 | N/A |
The substantial difference in boiling points indicates that zirconium remains in the solid or liquid phase much longer, which is vital for vacuum applications.
Is reactor safety better served by zirconium vs titanium alloys?

Reactor safety is uniquely dependent on zirconium because of its incredibly low thermal neutron absorption cross-section compared to titanium. In the specialized field of zirconium vs titanium for nuclear use, zirconium is the only viable choice for fuel cladding. This property allows neutrons to pass through the metal cladding to sustain the fission reaction without being “wasted” or causing the metal to become overly brittle.
Fuel element assembly structures
Zirconium alloys, such as Zircaloy, are used to house uranium fuel pellets within the reactor core. These assemblies must withstand intense radiation, high-pressure steam, and fluctuating temperatures. The best part? Zirconium’s corrosion resistance prevents radioactive leaks into the primary coolant loop.
Neutron absorption characteristics
By capturing very few neutrons, zirconium improves the fuel efficiency of the nuclear reactor. Titanium, conversely, has a much higher absorption rate, which would stifle the nuclear chain reaction. Here is the deal: without high-purity, hafnium-free zirconium, modern light-water reactors could not operate effectively.
- Neutron Transparency: Vital for maintaining the fission chain.
- Irradiation Stability: Resists swelling and cracking under flux.
- Hafnium-Free: Nuclear grade must have <0.01% hafnium.
Key Takeaway: For nuclear applications, zirconium is not just a preference; it is a fundamental requirement for the physics of power generation.
| Material | Neutron Cross Section (barns) | Role in Reactor | |
|---|---|---|---|
| Zircaloy-2/4 | 0.18 | Fuel Cladding | |
| Titanium | 6.1 | External shielding only | |
| Hafnium | 104 | Control Rods (Absorber) |
The contrast in neutron capture rates proves that zirconium is the superior engineering choice for core-adjacent components.
Which implants are superior when weighing zirconium vs titanium?
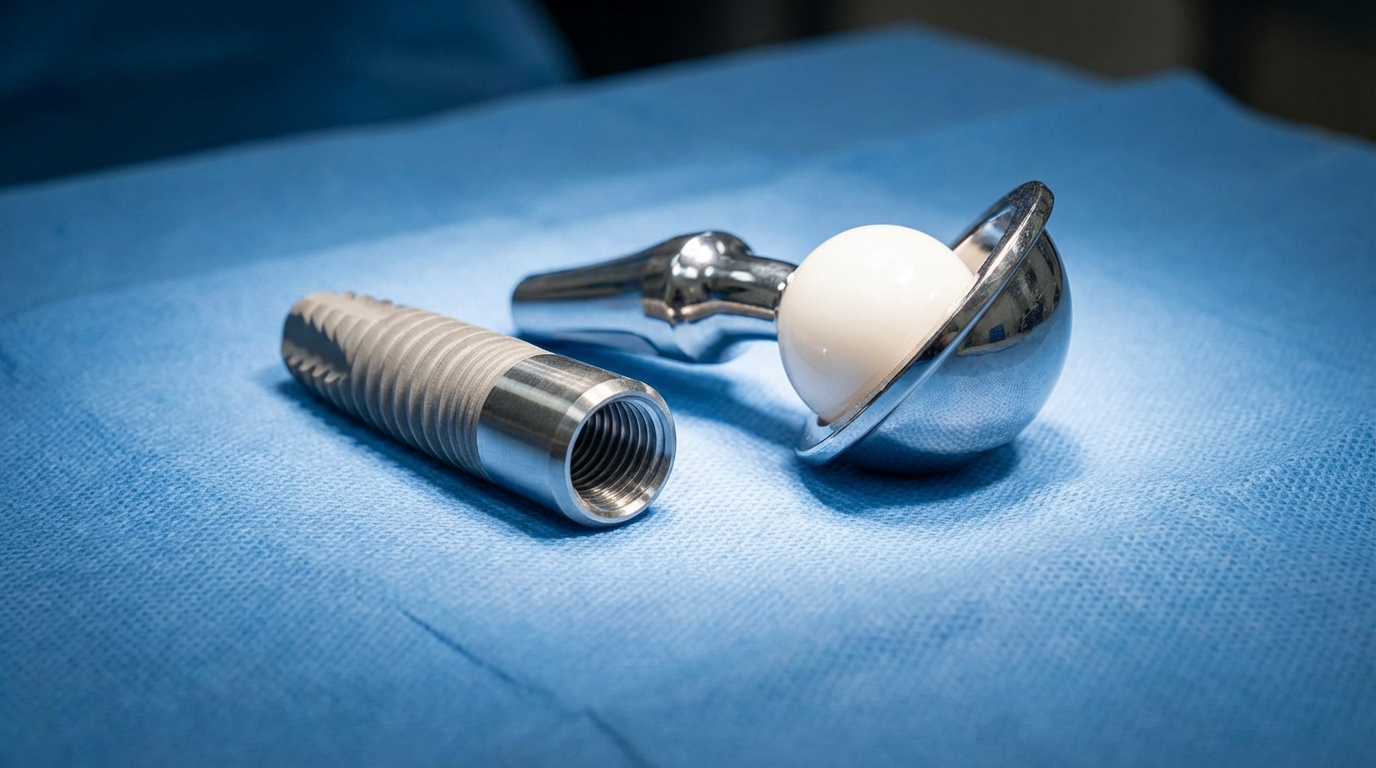
Zirconium dioxide (zirconia) is increasingly favored over titanium for dental and surgical implants due to its superior aesthetic match and complete biocompatibility. When doctors compare zirconium vs titanium , they often select zirconia for patients with metal sensitivities. Because it is a ceramic-like material in its oxide state, it does not conduct heat or electricity, reducing discomfort in oral applications.
Human tissue biocompatibility
Zirconium does not cause the inflammatory responses or allergic reactions that can sometimes occur with metal alloys. It integrates seamlessly with bone tissue, a process known as osseointegration. Think about it. This allows for long-lasting joint replacements and dental pins that feel like natural parts of the body.
Durability of dental crowns and pins
Zirconia crowns are virtually indestructible and can be color-matched to existing teeth with high precision. They offer high flexural strength, preventing the chipping that often plagues traditional porcelain-fused-to-metal restorations. Look at this: the smooth surface of zirconia also inhibits the growth of bacteria and plaque.
- Aesthetics: Hides the “dark line” at the gumline seen with metals.
- Conductivity: Non-conductive nature prevents galvanic shocks.
- Strength: Flexural strength exceeding 1200 MPa in 3Y-TZP grades.
Key Takeaway: Utilizing zirconium-based materials in medical device manufacturing ensures higher patient satisfaction and lower revision rates.
| Feature | Zirconia (ZrO2) | Titanium Alloy (Ti-6Al-4V) | |
|---|---|---|---|
| Biocompatibility | Exceptional | Very High | |
| Aesthetic Appeal | Natural Tooth Color | Metallic/Grey | |
| Conductive Properties | Non-conductive | Conductive | |
| Patient Sensitivity | Hypoallergenic | Possible metal allergy |
The move toward zirconia in modern dentistry reflects a broader trend toward biomimetic materials that prioritize both function and appearance.
What factors influence the cost of zirconium vs titanium metals?

The cost of zirconium vs titanium is influenced by the scarcity of high-purity ore and the extreme energy requirements of the Kroll reduction process. Zirconium is typically more expensive than titanium, with market prices ranging from 100 to 500 per kilogram depending on the purity and form. This pricing reflects the complexity of separating zirconium from its chemical twin, hafnium, which is present in almost all natural deposits.
Price trends and manufacturing cycles
Market fluctuations are often driven by the demands of the nuclear and aerospace industries. Because only a few countries possess a complete metallurgical cycle from concentrate to finished ingot, the supply remains relatively tight. Want to know more? The multi-stage purification including magnetic separation and air classification adds significant value to the final product.
Supply chain and availability factors
Procurement is often localized to major producers like Australia, China, and France. For you as a buyer, this means that lead times can be longer than for more common industrial alloys. However, the lifespan of a zirconium part often offsets the initial purchase price by reducing replacement frequency.
- Refining Costs: High electricity and chemical reagent usage.
- Scrap Recovery: Melting zirconium waste is a common way to lower costs.
- Nuclear Grade: High premium for hafnium-free material.
Key Takeaway: Evaluating the Total Cost of Ownership (TCO) rather than just the initial price will reveal zirconium’s true economic value.
| Factor | Impact on Zirconium Price | Impact on Titanium Price | |
|---|---|---|---|
| Ore Abundance | Trace element (High cost) | Abundant (Lower cost) | |
| Refining Steps | Complex (Hafnium removal) | Standard Kroll Process | |
| Industrial Scale | Medium | High | |
| Energy Consumption | Very High | High |
Understanding these cost drivers allows you to negotiate better terms and plan long-term project budgets with greater accuracy.
How does processing differ between zirconium vs titanium units?

Processing zirconium requires specialized atmosphere controls because the metal becomes highly reactive with oxygen and nitrogen at temperatures above 400°C. In the manufacturing of zirconium vs titanium components, vacuum or inert gas shielding is mandatory during welding and forging. If these precautions are ignored, the metal becomes brittle and loses its signature ductility.
Hot and cold processing techniques
Zirconium is highly ductile and amenable to forging, rolling, and stamping. It can be cold-worked significantly, but intermediate annealing in a vacuum is often necessary to relieve stress. Think about it. The ability to shape the metal at room temperature allows for the creation of intricate, thin-walled tubes for heat exchangers.
Impact of dissolved gas impurities
Small amounts of dissolved hydrogen or carbon can cause catastrophic failure in finished parts. Manufacturers use laser PSD analysis and XRF quality control to ensure that impurities are kept within strict limits. Here is the deal: maintaining a purity of 99.5% or higher is critical for maintaining fracture toughness in technical applications.
- Welding: Must be performed in a “glove box” or with trailing shields.
- Machining: Requires slow speeds and heavy feeds to prevent work hardening.
- Annealing: Vacuum furnace treatment is standard.
Key Takeaway: Partnering with a manufacturer that has in-house atmospheric control is vital to ensure the structural integrity of your zirconium parts.
| Processing Method | Required Conditions | Resulting Benefit | |
|---|---|---|---|
| Forging | Hot (with gas shield) | Refined grain structure | |
| Rolling | Cold or Warm | High surface finish | |
| Welding | Argon/Helium Purge | Ductile, leak-proof joints | |
| Annealing | Vacuum | Stress relief |
Careful management of these manufacturing parameters ensures that the final product meets the rigorous standards of the aerospace and chemical industries.
Why are ore sources critical for zirconium vs titanium output?

Ore sources are critical because the mineralogical composition of the zircon sand determines the feasibility of the entire metallurgical cycle. For the production of zirconium vs titanium, high-purity zircon concentrates (ZrO₂ + HfO₂ > 65%) are required to minimize the energy spent on impurity removal. Without a stable supply of these minerals, the production of metallic zirconium would be economically unviable.
Metallurgical cycles and trace elements
The journey from ore to metal involves heavy minerals like ilmenite and rutile being separated from zircon. Trace elements like iron (Fe₂O₃) and titanium (TiO₂) must be meticulously removed, especially for ceramic glazes where they can cause discoloration. It gets even better: companies like ZrBrontec maintain strict limits (TiO₂ < 0.1%) to ensure maximum whiteness in industrial applications.
Resource sustainability and rare-earth associations
Zircon is often found alongside rare-earth elements, making the mining process a complex multi-commodity operation. As global demand for clean energy grows, the extraction of these minerals must become more efficient and environmentally conscious. What does this mean for you? It means sourcing from ISO 9001:2015 certified manufacturers is the best way to ensure ethical and consistent material flow.
- Batch Consistency: Essential for predictable firing in ceramics.
- Traceability: COA-verified impurities (Fe, Ti, U, Th).
- Custom PSD: Matching particle size to specific manufacturing processes.
Key Takeaway: Your material reliability begins at the mine; choosing suppliers with vertically integrated production reduces your risk of batch defects.
| Mineral Property | Target Value | Impact on Metal Grade | |
|---|---|---|---|
| ZrO2 + HfO2 | ≥ 65% | High yield during reduction | |
| TiO2 Content | < 0.15% | Prevents color instability | |
| Fe2O3 Content | < 0.05% | Reduces magnetic interference | |
| Particle Size (D50) | 1–50 μm | Optimizes reactivity |
By focusing on these raw material specifications, you can ensure that your final metallic or ceramic products meet the highest global quality standards.
Strategic Conclusion
Industrial engineering is often a battle against the inevitable forces of corrosion and heat. Whether you are building a nuclear reactor core or a high-pressure chemical plant, the choice of materials defines your operational success. At ZrBrontec, we provide the high-purity zirconium silicate and zirconia materials that form the backbone of these critical industries. Our vision is to empower global manufacturers with the precision-engineered materials they need to innovate safely and sustainably. We invite you to experience the difference that 8,000 metric tons of annual capacity and a dedicated team of engineers can make for your project. To discuss your specific technical requirements or to request a quote for our high-whiteness zircon products, please contact us today .
Frequently Asked Questions
Can I use zirconium in environments where titanium has previously failed?Yes, zirconium is often the primary alternative when titanium suffers from pitting or general corrosion in highly concentrated hydrochloric acid or alkaline solutions. It provides a more stable oxide layer in these specific environments, significantly extending the service life of the equipment.
What is the best way to handle welding for zirconium components?You must use a strictly controlled inert atmosphere, such as argon or helium shielding, to prevent the metal from absorbing gases that lead to brittleness. Failure to provide adequate shielding will result in cracked welds and compromised structural integrity.
How do I know if I need nuclear-grade zirconium or industrial-grade?Nuclear-grade is required only if the metal will be used inside a reactor core where neutron transparency is vital, necessitating the removal of hafnium. For chemical processing, aerospace, and medical applications, industrial-grade zirconium (which includes hafnium) is the standard and more cost-effective choice.
Is it possible to color-match zirconia medical implants?Absolutely, zirconia can be manufactured in various shades to perfectly match the natural color of a patient’s teeth. This is why it is the preferred material for dental crowns and bridges where both strength and aesthetic appearance are required.
Can I replace traditional ceramic opacifiers with high-purity zircon?Yes, using high-purity zirconium silicate with a low TiO₂ content will provide superior whiteness and opacity in ceramic glazes. It helps hide surface defects and ensures a uniform finish that resists crawling and pinholes during the firing process.