Zircon is a naturally occurring mineral (ZrSiO4), while zirconium oxide is a synthetic advanced ceramic (ZrO2) engineered for superior performance in high-stakes industrial and medical environments. You are likely facing a critical material selection hurdle where terminology confusion leads to procurement risks and engineering failures. Choosing the incorrect material can result in premature component degradation, compromised safety, and significant financial losses during production. This analysis provides the technical clarity required to specify Zirconium Oxide with confidence, ensuring your application benefits from the highest standards of structural integrity.
Is Zirconium Oxide the same as natural zircon?

No, natural zircon and Zirconium Oxide are fundamentally different materials with distinct chemical identities and origins. Zircon is a silicate mineral extracted directly from the earth, whereas the oxide version is a highly refined ceramic produced through complex chemical synthesis.
Natural zircon crystals typically appear as brownish or translucent stones with a specific crystal structure. In contrast, the engineered oxide is a white, fine powder before it is sintered into its final, high-strength form.
Are the origins truly different?
You must distinguish between the raw geological source and the finished industrial product. While zircon serves as the primary ore, the extraction of Zirconium Oxide involves removing silicon and impurities to achieve high purity.
Check this out:
- Zircon is a “fine stone” used in jewelry and basic refractories.
- The oxide form is a technical ceramic for precision engineering.
- Zirconium is actually a metal, not a stone.
Key Takeaway: Use zircon for standard refractory applications and specify the oxide form for precision mechanical parts.
| Feature | Natural Zircon | Zirconium Oxide | |
|---|---|---|---|
| Origin | Natural Mineral | Man-made Synthetic | |
| Chemical Formula | ZrSiO4 | ZrO2 | |
| Primary Use | Jewelry/Refractories | Advanced Ceramics/Dental |
The geological path of a mineral is irrelevant once chemical processing transforms it into a high-performance ceramic.
What defines the chemistry of Zirconium Oxide?
Its chemistry consists of Zirconium and Oxygen atoms arranged in a crystalline lattice that excludes the silica found in natural zircon. This high-purity Zirconium Oxide structure allows for phase transformations that contribute to its legendary toughness and thermal stability.
Pure ZrO2 undergoes volume changes during heating, so engineers often add stabilizers like yttria or magnesia. This chemical modification creates “stabilized” versions that maintain their structural integrity across varying temperature ranges.
Why does molecular structure matter?
You will find that the absence of silicon atoms in the lattice significantly increases the material’s chemical resistance. This molecular refinement ensures that the ceramic remains inert when exposed to harsh industrial chemicals or bodily fluids.
Here is the kicker:
- The Zr-O bond is exceptionally strong and stable.
- Stabilizers prevent “crack propagation” through phase changes.
- Purity levels often exceed 99% in technical grades.
Key Takeaway: Chemical purity determines the lifespan of your components in corrosive or high-stress environments.
| Component | Zircon (ZrSiO4) | Zirconium Oxide (ZrO2) | |
|---|---|---|---|
| Silica Content | High (~33%) | Zero to Trace | |
| Purity Options | Industrial Grade | Up to 99.9% | |
| Phase Stability | Stable | Requires Stabilizers |
Molecular precision is the foundation of high-performance engineering in advanced ceramic manufacturing.
How hard is Zirconium Oxide on the Mohs scale?

Engineered Zirconium Oxide typically registers an 8.5 on the Mohs scale, making it significantly harder than natural zircon, which sits at approximately 7.5. This extreme hardness provides the material with exceptional resistance to scratching, surface wear, and mechanical abrasion.
The material’s hardness is paired with high fracture toughness, which is rare for ceramic materials. This unique combination allows Zirconium Oxide components to withstand impacts that would shatter standard industrial glass or porcelain.
Does hardness translate to durability?
You can rely on this material for applications where surface degradation would lead to total system failure. Its ability to resist abrasion means that dimensions remain stable even after thousands of operational cycles.
Look at this:
- Harder than steel and most natural gemstones.
- Resistant to chipping and surface cracking.
- Maintains a polished finish under heavy friction.
Key Takeaway: Choose the oxide form if your application involves high-friction contact or abrasive media.
| Material | Mohs Hardness | Fracture Toughness | |
|---|---|---|---|
| Zircon | 7.5 | Low | |
| Zirconium Oxide | 8.5 | High (8-10 MPa·m½) | |
| Diamond | 10.0 | High |
Hardness alone is insufficient; the combination of hardness and toughness defines the material’s mechanical superiority.
Why choose Zirconium Oxide for dental crowns?
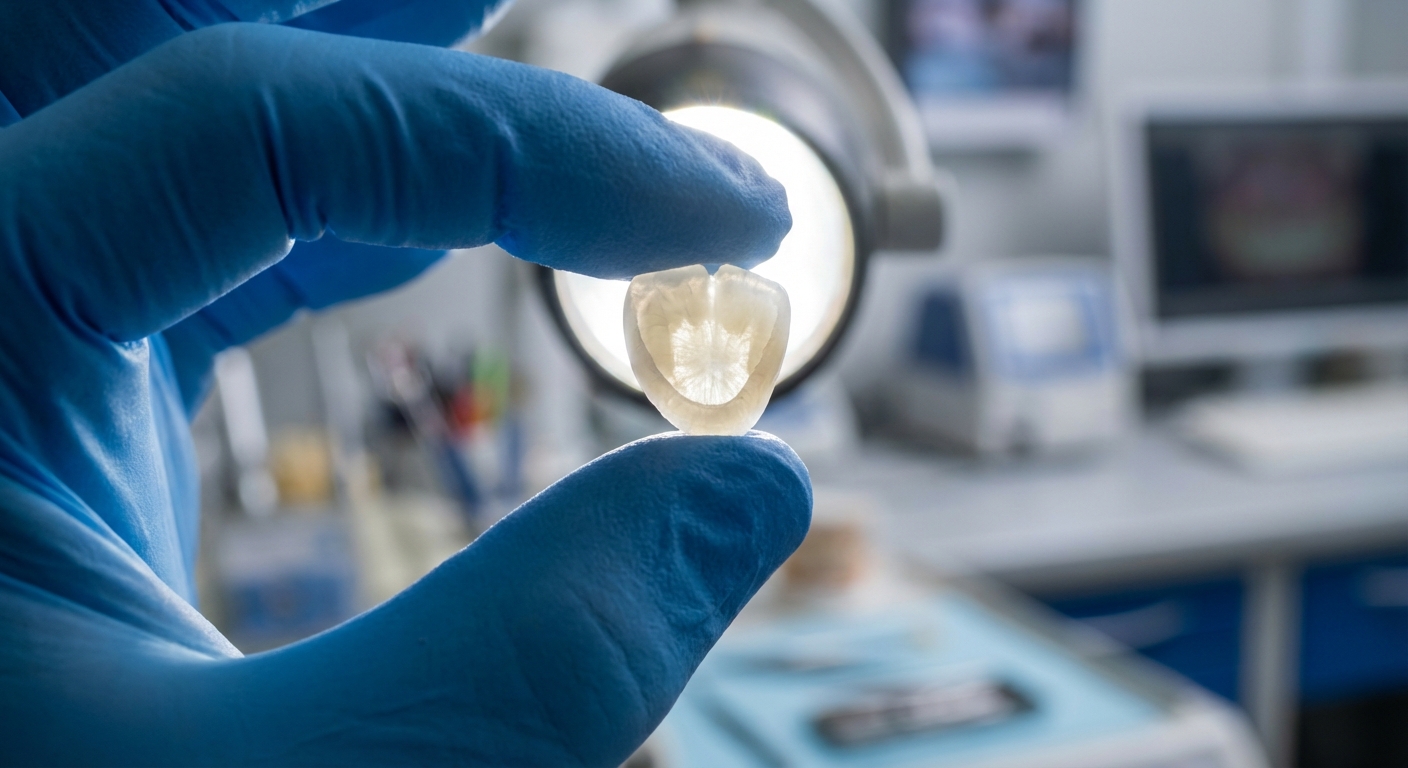
Dental professionals prefer Zirconium Oxide because it offers the perfect synergy of biocompatibility, extreme aesthetic translucency, and the structural strength required for permanent bridges. It has largely replaced traditional porcelain-fused-to-metal restorations due to its superior durability and natural appearance.
This material does not trigger allergic reactions and is chemically inert within the oral environment. Furthermore, Zirconium Oxide can be precision-milled using CAD/CAM technology to achieve microscopic accuracy for dental implants and abutments.
Is it better than traditional options?
You will notice that these crowns are virtually indistinguishable from natural tooth enamel while being significantly stronger. Their resistance to wear ensures that the restoration can handle the heavy forces of mastication over many years.
Think about this:
- Excellent light dispersion for a natural look.
- High bending strength prevents bridge fractures.
- Biocompatible nature supports healthy gum tissue.
Key Takeaway: For dental applications, prioritize Yttria-stabilized grades for the best balance of aesthetics and strength.
| Restorative Metric | Porcelain | Zirconium Oxide | |
|---|---|---|---|
| Flexural Strength | ~100 MPa | >1000 MPa | |
| Longevity | Moderate | Excellent | |
| Biocompatibility | High | Superior |
Modern dentistry relies on the structural reliability and biological safety provided by advanced oxide ceramics.
What are common Zirconium Oxide industrial uses?

In industrial settings, Zirconium Oxide is the material of choice for high-wear components like pump seals, valves, and precision bearings. Its chemical inertness makes it ideal for handling corrosive fluids in chemical processing plants where metals would quickly fail.
Manufacturers also utilize Zirconium Oxide for cutting tools, wire forming rollers, and extrusion nozzles due to its thermal stability. These parts maintain their geometry under extreme pressure and heat, reducing maintenance downtime.
Where else is it applied?
You can find this material in diverse sectors ranging from textile manufacturing to electronic sensor housing. Its versatility stems from its ability to be engineered into complex shapes with very tight tolerances.
Here is the deal:
- Precision ball valves for fluid control.
- Grinding media for high-efficiency mills.
- Insulating rings for high-temp electronics.
Key Takeaway: Replace failing metal parts with oxide ceramics to extend the service life of industrial machinery.
| Application | Benefit | |
|---|---|---|
| Pump Seals | Corrosion & Wear Resistance | |
| Cutting Tools | Edge Retention | |
| Bearings | Low Friction & High Load |
Industrial efficiency is maximized when the material properties are perfectly matched to the operational stress.
How is Zirconium Oxide synthesized from zircon?

The synthesis process involves the thermal decomposition of zircon sand followed by chemical leaching to isolate high-purity Zirconium Oxide. This transformation occurs in high-temperature kilns where the silica is stripped away, leaving behind the refined ceramic precursor.
Once the raw oxide is isolated, it undergoes micronization to control the particle size distribution. This ensures that the final Zirconium Oxide powder can be pressed and sintered into dense, void-free industrial components.
Is the refining process complex?
You should understand that achieving sub-micron particle sizes requires advanced milling technology and strict quality control. This precision during synthesis determines the final mechanical properties and the reliability of the sintered part.
But wait, there is more:
- Calcination temperatures exceed 1000°C.
- Chemical washing removes trace iron and titanium.
- Spray-drying creates flowable granules for pressing.
Key Takeaway: High-quality parts start with a synthesis process that ensures consistent particle size and phase purity.
| Process Step | Action | Outcome | |
|---|---|---|---|
| Decomposition | Heat ZrSiO4 | Separates Silica | |
| Leaching | Chemical Wash | High Purity | |
| Milling | Size Reduction | Sintering Reactivity |
Controlled chemical engineering is what turns common sand into an elite industrial material.
Can Zirconium Oxide handle high-heat stress?

Yes, Zirconium Oxide is an elite refractory material capable of withstanding extreme temperatures while maintaining low thermal conductivity. It is frequently used as a thermal barrier coating (TBC) on jet engine turbine blades to prevent metal fatigue.
In furnace applications, Zirconium Oxide serves as insulation and kiln furniture because it does not react with molten metals or glass. This thermal stability allows for higher operating temperatures, which directly improves the efficiency of industrial combustion processes.
Does it protect aerospace components?
You will find that TBCs made from this ceramic allow engines to run hotter than the melting point of the underlying superalloys. This capability is critical for modern aviation safety and fuel efficiency benchmarks.
The reality is this:
- Extremely low thermal conductivity (~2.2 W/m·K).
- High thermal expansion similar to steel.
- Resistance to thermal shock and cycling.
Key Takeaway: Use the oxide form for thermal barriers where heat shielding and structural stability are both required.
| Heat Metric | Metal Alloys | Zirconium Oxide | |
|---|---|---|---|
| Conductivity | High | Very Low | |
| Melting Point | ~1400°C | ~2700°C | |
| Heat Shielding | Poor | Excellent |
Thermal management in extreme environments is only possible through the application of advanced oxide barriers.
Is Zirconium Oxide more durable than gemstones?

In terms of chemical and mechanical durability, Zirconium Oxide far exceeds most natural gemstones and even its own natural counterpart, zircon. While gemstones are prized for optical beauty, the engineered oxide is designed for survival in the most aggressive chemical environments.
Unlike natural stones that might fracture under pressure or corrode in acid, Zirconium Oxide remains inert and structurally sound. It is a material that resists long-term degradation even when submerged in boiling concentrated acids or alkaline solutions.
Why compare them to gemstones?
You might encounter “cubic zirconia,” which is a fully stabilized form of this oxide used to imitate diamonds. While it looks like a gemstone, its industrial-grade cousins are much tougher and more resistant to mechanical stress.
Best of all:
- Immune to most industrial solvents and acids.
- Will not tarnish or degrade over time.
- Maintains structural integrity in salt spray.
Key Takeaway: If your environment is chemically hostile, the oxide form is your only viable long-term material choice.
| Resistance Type | Natural Zircon | Zirconium Oxide | |
|---|---|---|---|
| Acid Stability | Moderate | Excellent | |
| Scratch Resistance | High | Superior | |
| Longevity | Environmental Dependent | Decades in Industry |
Durability in engineering is defined by a material’s ability to remain unchanged by its surroundings.
Does Zirconium Oxide work well in sensors?

This material is an exceptional oxygen ion conductor, which is why Zirconium Oxide is the core component in automotive oxygen sensors and fuel cells. At high temperatures, the crystalline structure allows oxygen ions to move freely, enabling precise measurement of exhaust gases.
These sensors, often called lambda probes, rely on the ceramic’s ability to create a voltage difference based on oxygen concentration. Zirconium Oxide ensures that modern engines can optimize fuel injection and reduce harmful emissions significantly.
Are there other electronic uses?
You can also find this material in pressure sensors, sound transducers, and high-performance insulators. Its electrical resistivity combined with mechanical strength makes it a versatile substrate for advanced electronic packaging.
Keep this in mind:
- Critical for reducing vehicle emissions.
- Operates reliably in hot exhaust streams.
- Vital for solid oxide fuel cell (SOFC) technology.
Key Takeaway: Utilize the ion-conductive properties of the oxide for environmental monitoring and energy generation.
| Sensor Application | Role of ZrO2 | |
|---|---|---|
| Lambda Probe | Oxygen Ion Transport | |
| Fuel Cells | Solid Electrolyte | |
| Insulators | Electrical Isolation |
The ability to conduct ions while remaining a physical barrier is a rare and valuable engineering trait.
How to select the right Zirconium Oxide grade?

Selecting the correct grade of Zirconium Oxide depends on balancing requirements for strength, translucency, and the specific thermal conditions of your process. You must analyze the Particle Size Distribution (PSD) and phase composition (3Y, 5Y, or 8Y) provided in the manufacturer’s Certificate of Analysis (COA).
For high-load mechanical parts, yttria-stabilized tetragonal zirconia polycrystal (3Y-TZP) is usually the standard. However, if your application requires maximum heat resistance or optical clarity, you may need a fully stabilized cubic grade (8Y-FSZ).
What should you look for?
You need to verify that the supplier offers batch-to-batch consistency and full traceability from the raw ore to the finished powder. Inconsistent particle sizes can lead to uneven sintering and hidden structural weaknesses in your final components.
Now, consider this:
- 3Y grades offer the highest fracture toughness.
- 5Y and 8Y grades provide better translucency.
- Controlled PSD prevents warping during firing.
Key Takeaway: Always request a detailed COA to match the material’s specific characteristics to your engineering tolerances.
| Grade Type | Stabilizer % | Primary Advantage | |
|---|---|---|---|
| 3Y-TZP | 3 mol% Yttria | Max Strength/Toughness | |
| 5Y-PSZ | 5 mol% Yttria | Translucency/Esthetics | |
| 8Y-FSZ | 8 mol% Yttria | High-Temp Stability |
Selection success is the result of matching chemical specifications to the specific stress of the application.
Summary and Strategic Action
Distinguishing between natural zircon and engineered Zirconium Oxide is the first step toward achieving manufacturing excellence. While zircon is a valuable raw material, the oxide form is the powerhouse ceramic that solves the most difficult challenges in the dental, aerospace, and chemical industries. By understanding the nuances of hardness, chemistry, and phase stability, you ensure that your projects are built on a foundation of rigorous material science.
As a vertically integrated manufacturer, we specialize in bridging the gap between raw minerals and high-performance engineered components. We help you eliminate procurement risks by providing verified phase composition, controlled PSD, and full batch traceability. If you are struggling with component wear, thermal fatigue, or inconsistent sintering results, we can provide the technical guidance to optimize your material selection.
Ready to enhance your production yield with consistent materials? contact us today for a custom technical consultation and receive a grade recommendation tailored to your specific application.
Engineering high-stability zircon solutions is not just our business—it is our commitment to your industrial success.
Frequently Asked Questions
Can I use natural zircon for thermal barriers?No, you should not. While natural zircon has decent thermal properties, it lacks the extreme heat resistance and low conductivity of engineered Zirconium Oxide, which is required for critical aerospace and furnace shielding.
What’s the best way to clean oxide components?The best method is ultrasonic cleaning with mild solvents. Because the material is chemically inert, it resists most cleaning agents, but you should avoid highly abrasive pads that might eventually affect precision tolerances.
Is Zirconium Oxide safe for medical implants?Yes, it is extremely safe. It is highly biocompatible and does not react with bodily tissues, making it the preferred choice for long-lasting hip replacements and dental abutments.
How do I tell Zirconium Oxide from cubic zirconia?The best way is to check the application and grade. Cubic zirconia is the fully stabilized (8Y) gemstone version, while industrial zirconia refers to the high-strength (3Y) ceramic used for structural engineering.
Where can I buy high-purity ceramic grades?You should always buy directly from a manufacturer. A direct manufacturer provides full batch traceability and the technical data (PSD and COA) necessary to guarantee performance in industrial environments.